Recently, Jingxiang Su (first author), a postgraduate student in our laboratory, Professor Shixun Dai (corresponding author) and Ning Gan (Co-corresponding author) have published an article entitled “Optimized Ge-As-Se-Te chalcogenide glass fiber sensor with polydopamine-coated tapered zone for the highly sensitive detection of p-xylene in waters” in Optics Express (Optics Express. 2020, 28(1), 184-193 https://doi.org/10.1364/OE.381955). Reviewers highly value the innovation of this paper and consider it useful and important for the spectroscopy community, because it shows that further functionalization of the chalcogenide glasses (ChG) and fibers could lead to useful devices for the mid-IR region.
Organic pollutants are a class of high toxic, persistent and ubiquitous environmental contaminants that pose considerable risks to aquatic ecosystems and human health. The concentrations of most organic pollutants in real life are in the ppm or ppb level, which are lower than the previously reported detection limits in the field of ChG-tapered fiber (ChG-TF) sensing. Thus, exploring a novel ChG-TF sensor that can enable the detection of ppm or ppb concentration is essential. In order to improve the sensitivity of the ChG-TF sensor, we used a facile, in-situ self-polymerization method to generate a polydopamine membrane layer on the surface of the tapered zone of a Ge-As-Se-Te fiber. The sensitivity of the coated sensor is increased by 40 times and the detection limit is reduced to 50 mg/mL. The response time of the coated sensor is less than 10 min for the p-xylene aqueous solution in the concentration range of 50-2000 mg/mL, and the repeatability error is approximately 8.97%. Therefore, this novel ChG-TF sensor has great potential for online, in-situ environmental qualification.
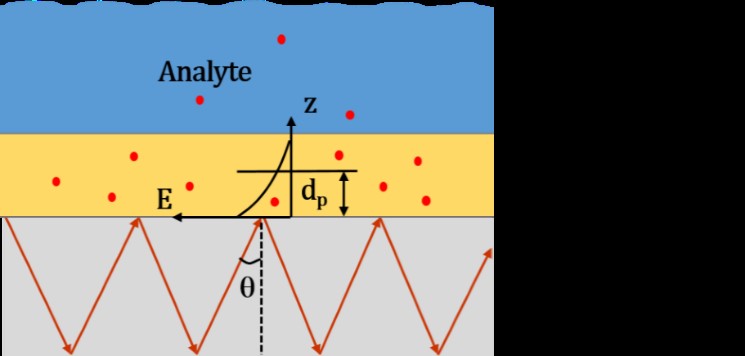
Fig. 1. General measurement principle of evanescent wave spectra of the polymer-coated fiber sensor (E, evanescent field; dp, penetration depth; q, angle of incidence).

Fig. 2. SEM images of the coated fiber (a) before and (b) after being immersed in an aqueous solution for 3h.
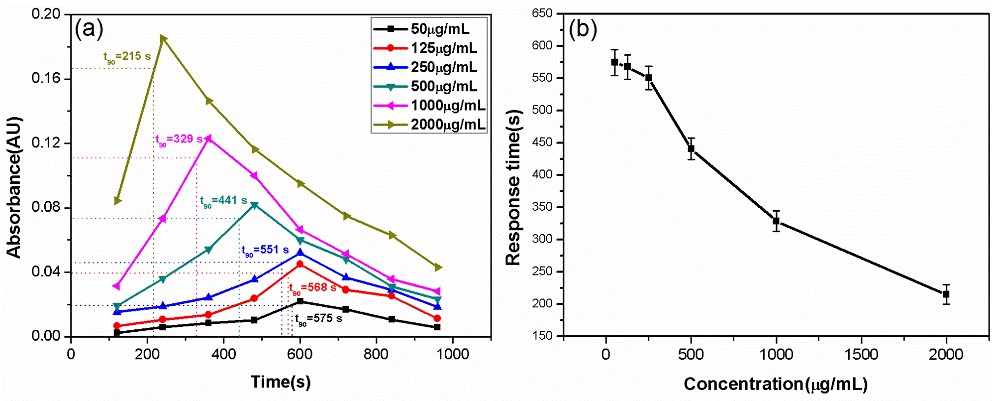
Fig. 3. (a) Relationship between the absorbance at the p-xylene absorption line and the time used for the polydopamine-coated ChG-TF sensor to sense six concentrations of the p-xylene aqueous solution. (b) Sensor response time varies with the p-xylene aqueous solution concentration for the polydopamine-coated ChG-TF sensor.
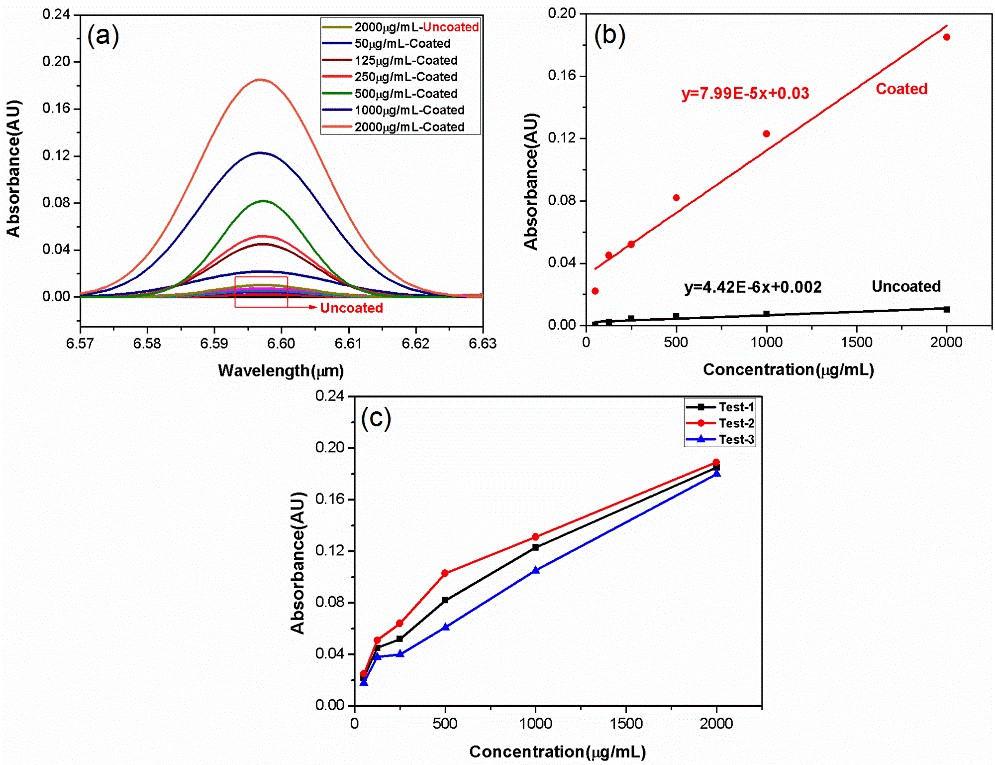
Fig. 4. (a) IR absorption spectra of six concentrations of the p-xylene aqueous solution recorded by coated and uncoated ChG-TF. (b) Dependence of the absorbance at the p-xylene absorption line on the p-xylene aqueous solution concentration for the coated and uncoated sensors. (c) Repeatability verification of the coated sensor under three tests.


